Digitization of clinical trials (Part 2)
Last time out we talked about how digitizing clinical trials has enabled incremental improvements to date — increasing the “power” of studies (the ability to derive conclusions) by encouraging compliance and collecting more data, and decreasing costs by leveraging of operational efficiencies. However, the impact of these changes has been limited by the online-offline gap — the fact that many clinical trial procedures still need to take place in-person. As we’ll get to, in-person visits are the single biggest reason why clinical trials are so slow and expensive. By taking them out of the equation, we’re able to truly leverage the scalability of digital technology and bring clinical trials into the digital age.
Part 2: Paradigm Change
Rethinking recruitment
Recruitment of eligible study participants is one of the biggest challenges with clinical trials today. In oncology, as one example, almost 20% of trials fail due to low recruitment. It’s also one of the biggest bottlenecks from a time perspective, as any delays in identifying potential study candidates will directly lead to a slower study.
There are many contributors to poor study recruitment, including the complexity of study protocols, insufficient marketing and communication, and the time burden for patients. Nevertheless, one of the biggest reasons is an obvious one, that the top of the funnel is too small. In other words, clinical trials simply aren’t reaching a large enough patient pool to find enough study candidates. Traditionally, clinical trials have operated using a site-centered model where clinical sites recruit patients through local outreach, by using their existing patient population or outbound marketing. The issue with this model is that even if you have a group of top-notch sites, you are still missing out on the vast majority of patients across the country who might qualify and be interested in your study if they knew about it.
The reason for the site-centered model is because screening patients for eligibility typically requires an in-person visit. Since it’s difficult to get patients to go out of their way for a screening visit, recruitment is limited to each clinical site’s geographic vicinity. If you imagine a study design, however, that could screen patients, collect data, and follow up with patients completely remotely, then we would be able to rethink recruitment from the ground up, and the top of our funnel would include all potential candidates across the country. This wouldn’t just be an incremental improvement on what we have today; it would be better by multiple orders of magnitude.
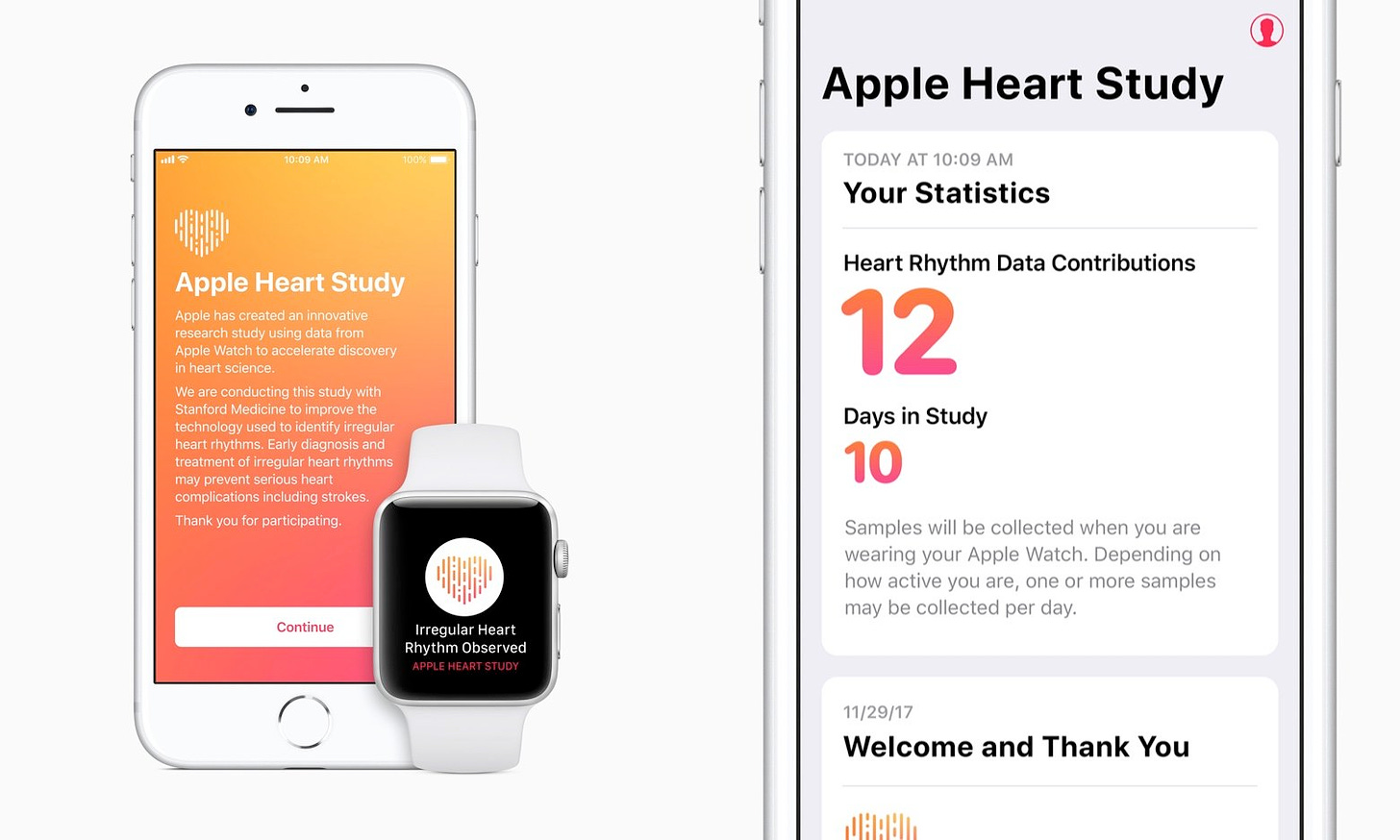
The most exciting proof-of-concept of this is Apple’s Watch Heart Study, which was published in NEJM last year. The goal of the study was to recruit participants with an iPhone and an Apple Watch, monitor their pulses over several months, and evaluate the ability of the Apple Watch to correctly identify cases of atrial fibrillation. All study procedures were conducted remotely, using telemedicine visits and digital data collection, and by mailing electrocardiography (ECG) patches to participants to use as a comparator. Data from this study served as the basis for the Apple Watch’s ECG app FDA approval in 2018 as a Class II medical device.
In this study, Apple was able to recruit a mind-blowing 419,297 participants in just 8 months, of which 2161 (0.52%) received notifications of irregular pulse. These kinds of numbers are simply unheard of for clinical research in any field, and would have been impossible if the study required in-person visits. Obviously, Apple was able to leverage their gigantic customer base to market the study, but these levels of recruitment would only be possible in a virtual clinical trial where patients could participate regardless of their geographic location.
Second-order benefits
Being able to recruit patients more quickly means completing studies faster, but it also means increased study power. In other words, all else equal, researchers are able to derive conclusions with greater confidence, and more easily weed out statistical noise. As we’ll touch on more later, this enables more flexibility around study design, and allows researchers to use outcome measures that might be less precise than the gold standard, but can be collected remotely. In other words, data from 100 patients on a gold standard outcome measure isn’t as useful as data from 1000 participants on a slightly less precise measure.
Virtual clinical trials also enable substantial cost savings by triaging patients more efficiently for follow-up while automating the remaining interactions. Apple’s Watch Heart Study would have been financially impracticable with a traditional budget — assuming only a single enrollment visit for each participant and nothing else (est. $75), the study would have cost $31.4m. Instead, the study automated the enrollment process and triaged patients so that only those who received irregular pulse notifications had a study visit.
Apple’s biggest market
It’s clear that virtual clinical trials come with many benefits, but you might be thinking that only a small fraction of conditions can be studied using fully virtual trials. You might be right, but I’d argue that this is the wrong way of thinking about it. The benefits of virtual clinical trials aren’t just incremental, they’re game-changing. The 10x (or 100x) improvements to time, cost, and study power give us the flexibility to figure out the other challenges, whether it’s using a less accurate outcome measure or recruiting for a rare disease. We should be envisioning virtual clinical trials as the endgame for every indication, and then overcoming the other challenges accordingly.
From what it looks like, Apple is already thinking along these lines. The Watch Heart Study is just the beginning, and Apple has already started 3 more similar studies: one focused on heart and movement, one on women’s health, and one on hearing. All of them share a couple of key similarities: (1) broad eligibility criteria (basically anyone can participate), and (2) primary endpoints that can be captured by Apple hardware (e.g. Apple Watch, AirPods, etc.) And for now, these are necessary; the former enables patients to be enrolled remotely, due to minimal screening requirements, and the latter allows for data to be collected remotely.
But what would it take for Apple to move into more complicated study designs, like those focused on a specific indication or intervention? The first challenge they would face is the screening problem, in other words, being able to identify potential study candidates without in-person visits. That’s where Apple Health Records could come in. If Apple is able to drive adoption of their Health Records system with both users and providers, they would have more than enough data on hand to screen patients for studies, or even match patients to studies, across a plethora of conditions. With Health Records, Apple may, intentionally or not, also be building the most powerful clinical trial recruitment engine in history.
The second challenge they would face is remote data collection beyond the measures that Apple devices are able to capture. On this front, mail-order health companies, like PillPack and Hims, provide a model to follow, whereby patients receive products and tests in the mail to be self-completed at home. In fact, Apple has already piloted this in their Watch Heart Study, by mailing ECG patches to patients who received notifications of irregular pulse and asking patients to wear them and return them for analysis. The quality of data self-collected by patients at home is bound to be worse, but as we mentioned earlier, the ability to recruit significantly more patients is more than enough to make up for the loss of precision.
Aggregation of clinical trials
We’ve talked before about how Stratechery’s aggregation theory is a useful framework for understanding the disruptive impact of digital technology, and how healthcare defies aggregation because of patients’ loyalty to providers and the need for in-person services. In many ways, the process of digitizing clinical trials mirrors this challenge; digital technology has improved the efficiency of research already, but its impact has been limited by the online-offline gap.
From the pioneering work of companies like Apple, we can see that while it would be impossible for medicine as a whole to go fully virtual, clinical trials can come pretty close. Although healthcare defies aggregation, clinical trials may not.